Newsletter
The latest newsletter includes what’s happening with monitoring, funding, nurseries, education, the calendar of events, and more. Looking for something older? View the newsletter archive. Receive the newsletter directly in your inbox by signing up for our mailing list.
Current California Oak Mortality Task Force Newsletter: May 2026
(Printable COMTF Report May 2026)
Regulatory Status Update
The changes to federal agencies brought by the actions of DOGE and other reorganization efforts have created interest in the current status of P. ramorum monitoring and regulatory programs. Outreach to two USDA programs related to P. ramorum—the USDA APHIS P. ramorum program and the USDA Forest Service national stream monitoring effort—uncovered answers to these questions.
USDA APHIS continues to maintain its national operations program of regulations enforcement, monitoring and diagnostics. Following is a summary of program activities in 2025.
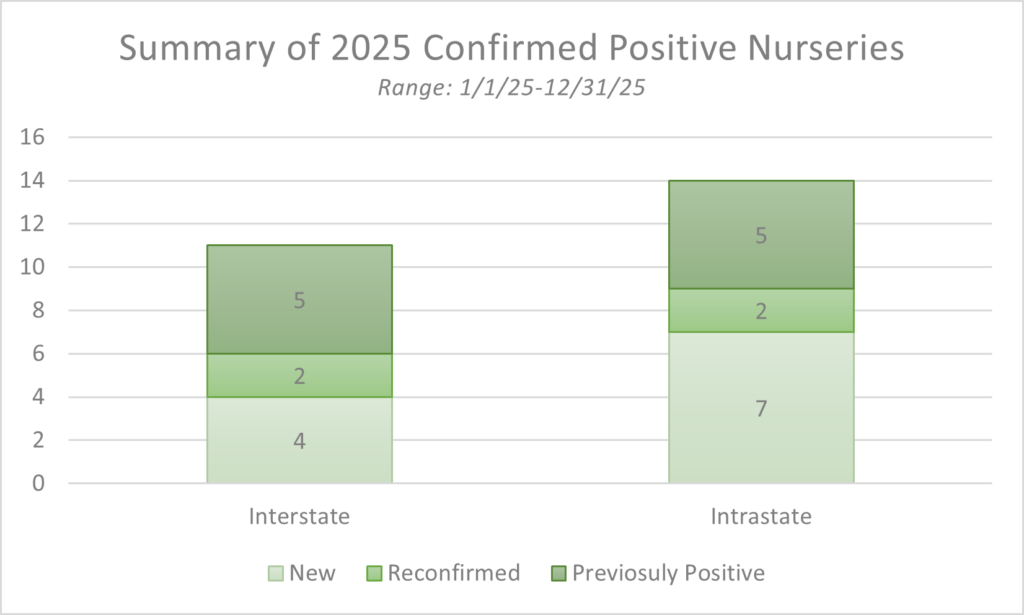
In 2025, the USDA Animal and Plant Health Inspection Service (APHIS), Plant Protection and Quarantine (PPQ) Phytophthora ramorum Program supported compliance activities, diagnostics, and surveys in 17 states. During 2025, confirmed positive detections of P. ramorum occurred at 24 nurseries across 7 states. Eleven were new locations, 10 were previously positive, and 4 were reconfirmed since they had a distant history in the program (Figure 1).
Of the 24 nurseries, 11 were interstate operations. Five of the nurseries were previously positive and in compliance agreements. Two nurseries were reconfirmed since they had a distant history with the program and will be reentering compliance. Four nurseries are new to the program and will be under compliance (Figure 1). One of the newly confirmed intrastate locations was both a nursery and a botanical garden.
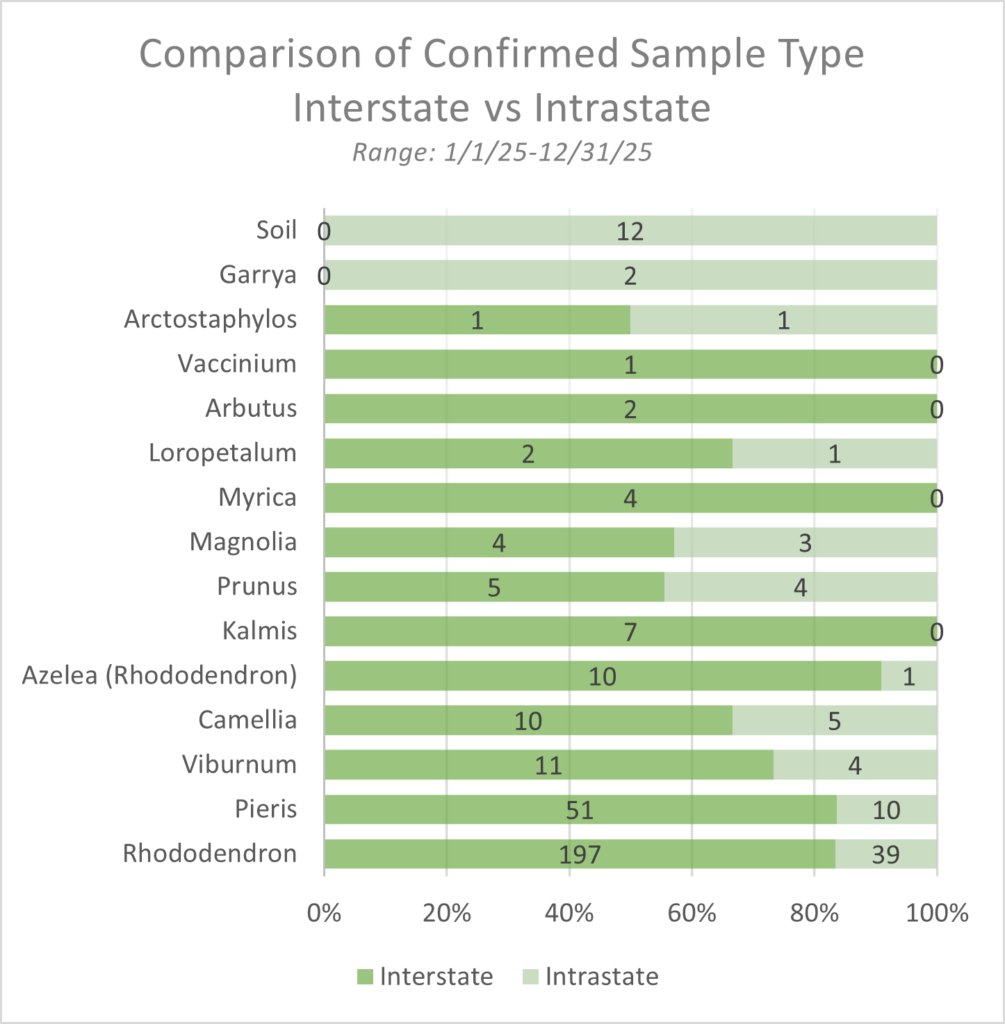
Confirmation of 11 new nurseries are a result of effective trace forward and trace back protocols. Nine trace investigations occurred, impacting 17 states and 1 international province. In 2025, 387 samples were confirmed positive across 14 genera (Figure 2). Since the last published P. ramorum Hosts and Associated Plants List (HAP) in 2022, there have been new species confirmed positive (Table 1).
Additionally, APHIS maintains an active policy program. For more information concerning national operations, contact Hannah Fritz, National Operations Manager, hannah.fritz@usda.gov. For more information about policy, contact Bill Wesela, National Policy Manager, william.d.wesela@usda.gov.
The USDA Forest Service will continue its national stream monitoring program, focusing mostly on watercourses in the eastern U.S., as separate university-coordinated stream monitoring programs take place on the West Coast. From mid-2024 through December 2025, the program processed nearly 600 samples from nine eastern states, and all were negative for P. ramorum. For information concerning the stream monitoring program, contact James Jacobs, St. Paul Field Office Forest Health Group Leader, james.j.jacobs@usda.gov.
Resources
A recording of the Webinar on Healthy Nursey Plants is now available. Over 500 participants joined the live event and the organizers are making the recording available to those who could not attend live. You can access the main part of the webinar on the PurPest Youtube Channel at https://youtu.be/5ASYxhOi_iY and the discussion at the end of the webinar is posted at https://youtu.be/GvsllDtkzTM.
Nurseries and Managed Landscapes
California Department of Agriculture P. ramorum Nursery Program report. Four California nurseries have had P. ramorum-positive plants so far in 2026. (1) In January, a previously P. ramorum positive production nursery in a quarantined county was again confirmed positive for P. ramorum after one Camellia plant was determined positive from a December 2025 inspection. No additional positive plants were detected during follow up delimitation inspections at that nursery. (2) After a trace back inspection for the previously mentioned positive Camellia plant detected at a quarantined county production nursery in January, another production nursery in a nonquarantine county was confirmed positive for P. ramorum on multiple varieties of Camellia plants. Trace investigations for this nursery are underway. (3) Another production nursery in a quarantined county was confirmed positive for P. ramorum on Rhododendron and Pieris species in January from samples collected in December 2025. During delimitation inspections, additional detections were made on Michelia, Pieris, and Rhododendron species. Trace forward inspections from this nursery detected a positive plant at a residence in a quarantined county. The quarantined county agricultural department is working with the homeowner on destruction of the positive plant at the residence. (4) A previously positive production nursery in a quarantined county was determined to be positive again in April after detecting P. ramorum on three Loropetalum chinense plants during a spring 2026 inspection. Trace investigations are ongoing.
All positive nurseries are undergoing the Confirmed Nursery Protocol from the USDA Phytophthora ramorum Domestic Regulatory Program Manual. Three of the positive nurseries are production nurseries in quarantined counties and one is a production nursery in a nonquarantined county. Plants confirmed positive for P. ramorum in 2026 are: Camellia sp. varieties ‘Debutante’, ‘Blood of China’, ‘Bob Hope’, ‘Cleopatra’, ‘Hana Jiman’, ‘Kramer’s Supreme’, ‘Nuccio’s Gem’, ‘Pearl Maxwell’, ‘Setsugekka’, ‘Silver Waves’, ‘Spellbound’, ‘Swan Lake’, and ‘White Doves’; Loropetalum chinense; Pieris sp., Pieris japonica ‘Bisbee Dwarf’; Michelia x foggii, Michelia sp. ‘Warm Fuzzies’; and Rhododendron sp., Rhododendron sp. ‘Alice Eastwood’, Rhododendron fragrantissimum ‘Coastal Spice.’ For more information, please contact Carolyn Lambert, carolyn.lambert@cdfa.ca.gov.
Washington State Department of Agriculture (WSDA) P. ramorum report. In January and February, WSDA conducted trace forward investigations on plants that shipped from two positive out-of-state nurseries. Follow up inspections and samples collected were negative for P. ramorum. In March, a positive Rhododendron was detected in the quarantine hold area at a botanical garden in Kitsap County. Garden staff hold and monitor incoming host plants for symptoms of P. ramorum prior to introduction into the garden, and contact WSDA when they see something suspicious. Twice in the past year, the quarantine hold area has functioned as intended. A positive Garrya was detected last November. In the March incident, all plants in the quarantine hold area were destroyed, and all perimeter samples were negative. WSDA is conducting a trace-back investigation on the positive Rhododendron. For more information, contact Scott Brooks, SBrooks@agr.wa.gov.
Research
Editorial comment: As we saw with the 2025 paper by Frankel, et al., there is a risk in the naïve use of open-source data. This issue has recently appeared again. As such, we would remind readers that this Research section and the associated P. ramorum Bibliography linked on the COMTF website do not capture every article related to Phytophthora or even P. ramorum. We will prioritize articles from known and established scientists and academic journals, and you are welcome to conduct your own Google Scholar search or literature review to fill in any gaps.
Mainello-Land, A., Saville, A.C., Patrick, C., Harris, A., Van Poucke, K., O’Hanlon, R., and Ristaino, J. 2026. Genetic Structure and Migration Routes of Phytophthora ramorum into Ireland and Northern Ireland. Phytopathology. 2026 Mar 03. doi:10.1094/PHYTO-10-25-0346-R.
Abstract: Phytophthora ramorum is a plant pathogen that causes disease in nurseries and forests in the United States, Canada, Asia, South America, and Europe. Since 2002, disease caused by P. ramorum has led to the destruction of horticultural plants and trees in Ireland (IE) and Northern Ireland (NI). Two lineages, EU1 and EU2, have been reported in both countries; EU1 is more prevalent in IE, while EU2 is more prevalent in NI. We examined the genetic structure of P. ramorum populations in IE and NI from 2005-2022 using microsatellite genotyping. Populations were sorted by country, county, lineage, year, host, and source (nursery or forest). Twenty-nine EU1 multilocus genotypes (MLGs) and six EU2 MLGs were observed. The EU1MG1, the most prevalent MLG in both countries (64% IE, 57% NI), was first observed in nurseries on rhododendron and later found in forests on larch. The EU2MG1 was more common in NI, while EU2MG3 was more common in IE. By 2006, four EU1 MLGs were identified from nurseries and forests in IE suggesting P. ramorum had already been introduced multiple times, likely via the plant trade. Minimum spanning networks revealed similar EU1 MLGs in IE nurseries and forests, and in forests in IE, the UK, and Europe. A T-BAS SSR interactive phylogenetic tool was created to query the EU1 and EU2 lineages. Our data place IE and NI P. ramorum populations into a broader European context and revealed novel genotypes of P. ramorum EU1 and EU2 in IE and NI.
Pham, D.H., Pastalka, T., Liu, H., and Schwigkofler, W. 2026. First report of Phytophthora ramorum causing leaf blight on Nerium oleander in the United States. APS Online: 24 Feb 2026. https://apsjournals.apsnet.org/doi/10.1094/PDIS-12-25-2455-PDN.
Abstract: Nerium oleander L. (Apocynaceae; common oleander) is a woody plant native to the Palearctic and planted as an ornamental worldwide. In May 2024, small brown necrotic spots were detected on leaves of a residential oleanders, an area with many California Bay Laurel (Umbellularia californica) infested by P. ramorum, in Mill Valley, Marin County, CA (37.90481ºN, 122.55282ºW). Symptomatic leaves tested positive using a genus-specific lateral flow assay (LFA) for Phytophthora (Agdia Inc, Elkhart, IN, USA). Leaves were surface sterilized using 70% ethanol, plated on selective PARPH media (Jeffers et al. 1986) and incubated at 20ºC. After 6 days, a single culture was isolated (NORS074) from the necrotic spots and produced morphological structures typical of P. ramorum, including coralloid mycelium, chlamydospores and semi-papillate sporangia (Fig. 1) (Werres, S., et al. 2001). The internal transcribed spacer (ITS) and beta tubulin regions were sequenced using the primers ITS1/ITS4 (White et al. 1990; accession no. PX270955) and TUBUF2/TUBUR1 (Kroon et al. 2004; accession no. PX551880), respectively. A BLAST search revealed 99.50% identity for ITS and 99.68% for the beta tubulin with P. ramorum ex-type strain CBS 101553 (accession no. NR_147877.1 and LC595884.1, respectively). Ten unwounded and ten wounded leaves of N. oleander were inoculated with strain NORS074 using two methods: i) mycelial plugs and ii) zoospore solution (1.72 x 105 zoospores/ml). All four treatments induced necrotic leaf spots; and P. ramorum was re-isolated from the leaves. Sporulation of the isolate on inoculated leaves was observed. Pathogenicity tests were also performed on whole plants of N. oleander variety Austin City Limits (N = 10; 40 cm tall and 35 cm wide) using two methods: i) a zoospore solution (1.9 × 105 zoospores/ml) was sprayed on the plants until runoff; ii) leaf tips were immersed in 1.5 ml of the zoospore solution in a 15 ml conical tube attached to the plant during entire experiment. Ten leaves per plant were wounded for each assay. The inoculations were carried out in an open-field environment to simulate natural condition. Plants were misted with water and kept individually in plastic bags for 72 hours to maintain humidity level. Leaves inoculated with all four different treatments developed symptoms 12 dpi consisting of small black necrotic spots and lesions along the midrib vein (Fig. 2). Symptomatic leaves tested positive using the same Phytophthora LFA, and P. ramorum was re-isolated on PARPH media. The identity of the re-isolates was confirmed morphologically and by ITS sequencing. No symptoms were detected on untreated control plants. To our knowledge, this is the first report of P. ramorum occurring on N. oleander in the United States, and the first time ever, that Koch’s postulates have been confirmed for this host-pathogen combination. Previously, P. ramorum was detected on N. oleander in Ireland, but no inoculation studies were reported (O’Hanlon et al. 2016). This study provides the evidence to reclassify N. oleander from an APHIS ‘associated host’ (APHIS 2022) to a proven host. An ‘associate host’ is a plant from which a pathogen was isolated; a ‘proven host’ is a plant, for which Koch’s Postulates were confirmed. The original infection, occurring during the exceptionally wet 2023/24 winter in a shaded area, was condition-dependent; failure to re-isolate P. ramorum in Spring 2025 suggests the occurrence was transient and driven by extreme environmental pressures.
Related Research
EFSA Panel on Plant Health (PLH). 2026. Commodity risk assessment of Hamamelis mollis plants from the UK. EFSA J. 2026 Feb 24;24(2):e9943. doi:10.2903/j.efsa.2026.9943.
Abstract: The European Commission requested the EFSA Panel on Plant Health to prepare and deliver risk assessments for commodities listed in Commission Implementing Regulation (EU) 2018/2019 as ‘High risk plants, plant products and other objects’. This Scientific Opinion covers plant health risks posed by rooted plants in pots of Hamamelis mollis imported from the United Kingdom (UK). The assessment was performed considering the available scientific information, including the technical information provided by the UK. All pests associated with the commodities were evaluated against specific criteria. Two EU regulated pests (Phytophthora ramorum non‐EU isolates and Scirtothrips dorsalis), present in the UK and potentially associated with the commodity, were considered as relevant for this Opinion. No pests non‐regulated in the EU were identified to be selected for further evaluation.
EFSA Panel on Plant Health (PLH). 2026. Commodity risk assessment of Robinia pseudoacacia plants from the United Kingdom. EFSA J. 2026 Apr22;24(4):e10056. doi:10.2903/j.efsa.2026.10056.
Abstract: The European Commission requested the EFSA Panel on Plant Health (PLH) to prepare and deliver risk assessments for commodities listed in Commission Implementing Regulation (EU) 2018/2019 as ‘High risk plants, plant products and other objects’. This Scientific Opinion covers plant health risks posed by rooted plants in pots of Robinia pseudoacacia ‘Frisia’ imported from the United Kingdom (UK). The assessment was performed considering the available scientific information, including the technical information provided by the UK. All pests potentially associated with the commodity were evaluated against specific criteria. Two EU‐regulated pests (Bemisia tabaci and Phytophthora ramorum non‐EU isolates), present in the UK and potentially associated with the commodity, were considered as relevant for this Opinion. No non‐regulated pests in the European Union were identified to be selected for further evaluation.
EFSA Panel on Plant Health (PLH). 2026. Commodity risk assessment of Salix unrooted cuttings from the UK. EFSA J. 2026 Feb 23;24(2):e9947. doi:10.2903/j.efsa.2026.9947.
Abstract: The European Commission requested the EFSA Panel on Plant Health to prepare and deliver risk assessments for commodities listed in Commission Implementing Regulation (EU) 2018/2019 as ‘High risk plants, plant products and other objects’. This Scientific Opinion covers plant health risks posed by unrooted cuttings of Salix species (S. aegyptiaca, S. eriocephala, S. gmelinii, S. miyabeana, S. purpurea, S. rehderiana, S. schwerinii, S. udensis, S. viminalis) imported from the United Kingdom (UK). The assessment was performed considering the available scientific information, including the technical information provided by the UK. All pests potentially associated with the commodities were evaluated against specific criteria. Only two EU Regulated pests (the fungus Entoleuca mammata and the oomycete Phytophthora ramorum (non‐EU isolates)), present in the UK and potentially associated with the commodity, were considered as relevant for this Opinion. No pests non‐regulated in the EU were identified to be selected for further evaluation.
Papini, V., Benigno, A., Rizzo, D., & Moricca, S. 2026. Catching the Elusive Phytophthora: A Review of Methods and Applications for Pathogen Detection and Identification Across Agricultural, Horticultural, Forestry and Ornamental Settings. BioTech 15(1), 17. https://doi.org/10.3390/biotech15010017.
Abstract: Species of the genus Phytophthora are among the most detrimental plant pathogens globally, representing a significant threat to global agriculture, horticulture, and forestry. These zoosporic oomycetes have historically caused devastating outbreaks, including, just to mention a few, late blight of potato in Ireland; jarrah dieback of eucalyptus in Western Australia; ink disease of chestnut in Europe; sudden oak death and sudden larch death of coast live oak and tanoak in the Western US, and of Japanese larch in the UK. The environmental and ecological impacts of the diseases they cause result in significant economic costs that often have social repercussions. With the acceleration of globalization, enhancing the movement of plant material, in particular with the global live plant trade, the spread of Phytophthora to new, uncontaminated territories has intensified. Nurseries play a key role in the movement of these pathogens, the trade of contaminated stocks representing their major dissemination route. However valuable, conventional detection techniques, including baiting and direct isolation, are too slow and labor-intensive to meet current diagnostic requirements, particularly given the huge volumes of plants traded globally. This problem becomes even more acute when large volumes of potentially infectious plant material need to be processed in a short time frame, as it is often necessary to provide accurate and timely responses to interested parties. Early and precise detection is thus vital to avert outbreaks and mitigate long-term consequences. This review evaluates and contrasts the efficacy of novel detection methods against traditional approaches, emphasizing their significance in managing the escalating threat posed by Phytophthora spp. worldwide. Despite technological advances, critical challenges remain that limit the reliability and large-scale adoption of new diagnostic methods. Research still needs to bridge the gap between the laboratory and the field in terms of accuracy, sensitivity and diagnostic costs. Recent innovations focus on sensor technology and point-of-care (POC) devices for faster, more sensitive, and low-cost specific detection of Phytophthora spp. in plant matrices, water and soil. Enhancing diagnostic capabilities through these tools is crucial for protecting agricultural productivity, local economies, and natural ecosystems.



